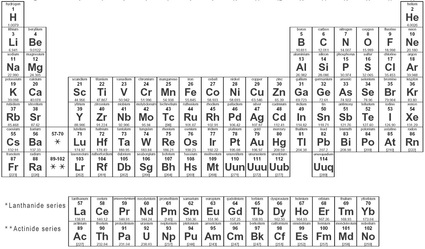
Elements
The periodic table is composed of many squares that each represent a different element. The amount of information given in each square can differ between periodic tables, but each element is always in the same place. Information that is commonly given by most periodic tables includes the symbol, the name, the atomic number, and the atomic mass.
Symbol
Each element has its own unique symbol that consists of a single capital letter or one capital and one lower case letter. The symbol for each element sometimes relates to the letter that the element's name begins with, but this is not always the case, such as in the two examples below.
The atomic number represents the number of protons within the nucleus of an element. The number of protons in the nucleus is different for every element but is the same for every atom within one element. In the examples above, the atomic number of hydrogen is 1. This means that any element that has one proton is hydrogen and every atom of hydrogen has only one proton. Potassium contains 19 protons within the nucleus.
Atomic Mass
The atomic mass of an element on the periodic is an average value. The mass of an atom is calculated by adding the number of protons in the nucleus to the number of neutrons in the nucleus. Since the number of neutrons can vary between atoms of the same element, the atomic mass of an element is not consistent so the average value is given on the periodic table. One can use the atomic mass to find the most common number of neutrons within an element. In the examples above, one can find the number of neutrons by subtracting the atomic number from the atomic mass, so
Atomic Mass - Atomic Number = # of Neutrons
For hydrogen this would be
1-1= 0 Neutrons
And for Potassium it would be
39-19= 20 Neutrons
Atomic Mass - Atomic Number = # of Neutrons
For hydrogen this would be
1-1= 0 Neutrons
And for Potassium it would be
39-19= 20 Neutrons
Current Research
Scientists have recently confirmed the existence of element 115. In 2013, a group of Swedish researchers created an element with 115 protons in its nucleus by smashing elements together. The same element was created by Russian researchers in 2004. The group of Swedish scientists confirmed the existence of the element. The confirmation of the elements existence means that it will appear on periodic tables created after 2013. The last element on the periodic table that appears in nature is Uranium with 92 protons. Every element on the periodic table after Uranium has been synthesized in labs.
http://www.cnn.com/2013/08/28/world/europe/new-chemical-element/
http://www.cnn.com/2013/08/28/world/europe/new-chemical-element/
As recently as early this year, scientists at the GSI Helmholtz Center for Heavy Ion Research in Germany have reportedly created element 117 in an accelerator laboratory. The element is currently unnamed (it has been temporarily been named ununseptium) but is making progress towards becoming an official part of the periodic table. Element 117 is a super-heavy element that is not found in nature. The element can only been synthesized in laboratories by scientists. Element 117 along with all other super-heavy elements above the atomic number 102 are part of scientific research to discover just how heavy atoms can get. The recent recreation of the element by German scientists, which followed the original discover of the element by a team of American and Russian scientists in 2010, prompted the International Union of Pure and Applied Chemistry (IUPAC) to review the discover and decide whether or not to formally include element 117 on the periodic table.
http://www.foxnews.com/science/2014/05/05/new-super-heavy-element-117-confirmed-by-scientists/
http://www.foxnews.com/science/2014/05/05/new-super-heavy-element-117-confirmed-by-scientists/
Other Resources for Elements of the Periodic Table
Explanatory Video
Web Resources
http://chemwiki.ucdavis.edu/Physical_Chemistry/Atomic_Theory/Nuclide,_Atomic_Number,_mass_number
http://nuclearpowertraining.tpub.com/h1015v1/css/h1015v1_26.htm
http://nuclearpowertraining.tpub.com/h1015v1/css/h1015v1_26.htm
Image Sources:
http://sciencenotes.org/wp-content/uploads/2013/05/01-Hydrogen-Tile.png
http://sciencenotes.org/wp-content/uploads/2013/05/01-Hydrogen-Tile.png